Booze, Brains, and Bubble Goo
Why alcohol is called spirit and how it serves the humankind
SCIENCEHISTORY
3/3/20257 min read


“All alcohols are born equal, but some alcohols are more equal than others.”
(Yes, George Orwell never partied with polyols, but he'd get the gist.)
Picture this: You're at a bar, sipping what you think is the only kind of alcohol worth knowing — ethanol, the whiskey sibling that turns you into a temporary poet (or fool). But hold your glass! In the chemist’s world, “alcohol” is a whole riotous family of molecules, all wearing at least one -OH (hydroxyl) group like a party hat. Methanol, ethanol, isopropanol, ethylene glycol, glycerol — they’re all alcohols, yet they behave wildly differently. Some burn with yellowish flames and kill silently; some give you a buzz; others are sweet and useful; and a few are downright heroic. Let’s meet them, buckle up — these molecules have personalities!
Why Do They All End in “-ol”?


The word alcohol actually started as “al-kuḥl,” an Arabic term for a fine powder used as eyeliner (kohl). How come the eyeliner and the booze are the same? It appears that about 2000 years ago Egyptians knew to “distillate” mineral stibnite. This gem, in difference with the other stones, does not melt into a liquid while being heated. It becomes a “vapor” immediately, so called “sublimes”. And when the stibnite vapor cools down, it forms so tiny black crystals, so fine powder that it can be used as eyeliner. It was also used as antiseptic. In ancient times people believed that sublimized stone kept its “spirit” or essence in the powder. This is why 1700 years ago, 3 centuries after the stibnite sublimation, the discovered process of the wine distillation resulted with “alcohol” or “spirit”.
Over centuries, it shifted to mean any very pure, distilled substance, and finally, in chemistry, it came to mean any organic compound with one or more hydroxyl (-OH) groups attached to a carbon chain. That’s why we call them:
methanol (one carbon, one -OH)
ethanol (two carbons, one -OH)
isopropanol (three-carbon chain, -OH in the middle)
ethylene glycol (two carbons, two -OHs)
glycerol (three carbons, three -OHs)
The suffix “-ol” is a chemical shorthand: wherever you see it, there’s an -OH group begging to form hydrogen bonds, dissolve in water, or react with acids, oxidants, and more. All alcohols share that -OH swagger, but the rest of the molecule decides whether it’s poison, party fuel, or skin food.
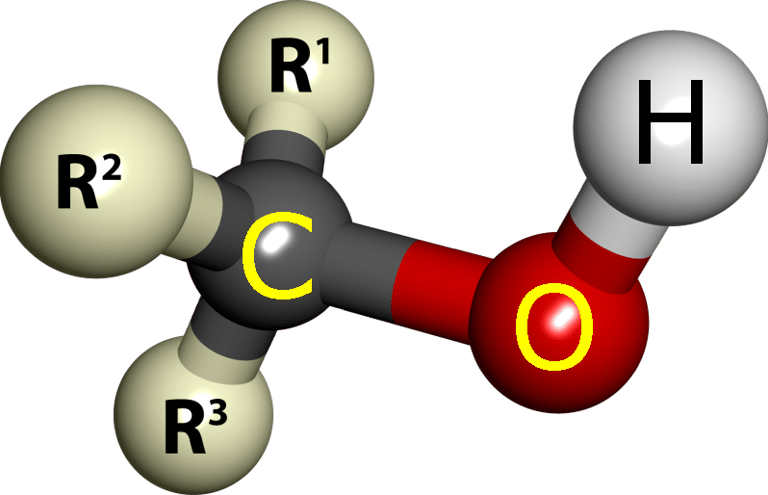
The Solo Sippers: Monoalcohols (One -OH Group)
These lone-wolf alcohols pack a punch with just one hydroxyl (-OH) group. They're small, feisty, and love messing with flames and your insides.
Methanol (CH₃OH): The goth cousin who stinks and burns with a spooky yellowish flame. Physically? It's a colorless liquid that boils low (65°C), slipping into your bloodstream like a ninja. Reaction-wise, it oxidizes to formaldehyde (nasty stuff for pickling brains, not yours). Verdict: Lethal poison—blindness (just from 10ml at vodka concentration) or death in small sips (150-200 ml at 40% will kill you with probability 50%). Steer clear unless you're convinced you saw enough in this life, literally.
Ethanol (C₂H₅OH): Party hero! Blueish flame on the barbie, boils at 78°C, and mixes with water like old pals (fully miscible). Your liver turns it into acetaldehyde (temporary hangover toxin), then harmless acetate. It's the "equal" one—fun in moderation, but overdo it and you're singing bad karaoke. Used in everything from booze to hand sanitizer. Funny fact, ethanol, being also a venom, is an antidote for poisoning with methanol. They both are landing at the same “airfield”. So whoever of them is bigger wins.
Isopropanol ((CH₃)₂CHOH): The rubbing alcohol champ, with a ketonic (not pleasant) whiff. Boils at 82°C, less toxic than methanol but still no cocktail mixer. It dehydrates via oxidation to acetone (nail polish remover vibes). Physical quirk: Evaporates fast, cooling your skin like a minty breeze. Great for wiping screens, not swigging.
Monoalcohols? Volatile flyers — low boiling points, flammable, and they love forming azeotropes (stubborn mixes with water that distillation can't split easily).
Double Trouble: Diols (Two -OH Groups)
Enter the twosome! These have two -OH crews, making them stickier and sweeter. Higher boiling points (hello, hydrogen bonding overload) mean they're less evaporate-y.
Ethylene glycol (HO-CH₂-CH₂-OH): Antifreeze king — sweet-tasting liquid that freezes at -37°C and boils at 197°C. Physical peculiarity: Lowers freezing point of water (perfect for car radiators in snowy Piedmont winters). Reaction star: Polymerizes into PET plastic (your soda bottles). But beware—dogs lap it up thinking it's candy, then kidney shutdown. Toxic, but useful!
Diols hug water tighter, making them hygroscopic (moisture magnets) and ideal for lotions or de-icers.
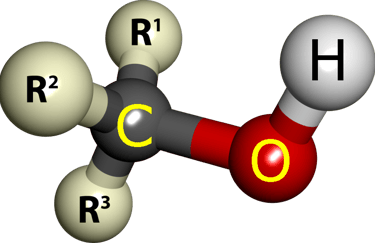
Triple Threat: Triols (Three -OH Groups)
Now we're talking sweet overachievers! Glycerol leads the polyol parade—viscous, non-volatile syrups that laugh at low temps.
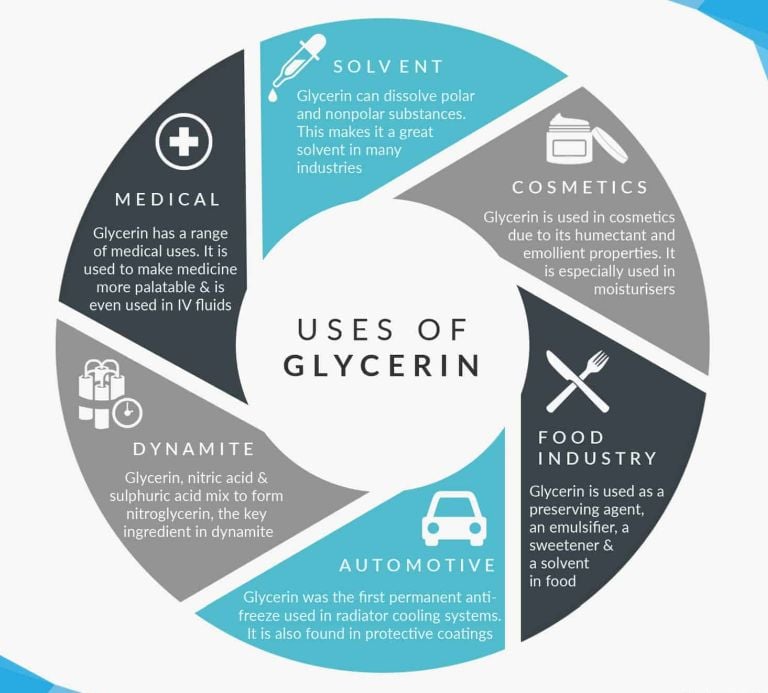
Glycerol (or glycerin, C₃H₅(OH)₃): The good guy! Thick as honey (boils at 290°C, won't budge), mixes with water forever, and sweetens without calories. Physical quirk: Humectant superstar — pulls moisture into skin (lotion magic) or keeps cakes moist. Reactions? Esterifies into soaps or nitroglycerin (boom potential, handled carefully). Found in foods, meds, and even your blood (as triglycerides). Not bad — it's a hero!
Gut-Friendly Polyols: Sweet, but Play Fair
Polyols like sorbitol or xylitol? Sugar alcohols in gum — sweet, low-cal, and gut-friendly (mostly). They form gels, sweeten without spikes, and prove alcohols can be your gym buddy, not your downfall.
You’re right to be cautious: yes, in very large amounts, sugar alcohols like sorbitol, xylitol, and maltitol can cause gas, bloating, and diarrhea, because they aren’t fully absorbed in the small intestine and pull water into the gut (osmotic effect). But in sensible quantities, they play a useful role:
Better for teeth: Unlike sucrose, polyols aren’t fermented by the bacteria that cause cavities, so they’re “non-cariogenic” (tooth-friendly).
Gentler on blood sugar: They have a low glycemic index, so they don’t spike insulin, making them useful for people with diabetes or prediabetes.
Prebiotic-like effect: Some polyols (like xylitol, lactitol, and isomalt) reach the colon, where they can feed beneficial bacteria like Bifidobacteria and promote the production of short-chain fatty acids, which may support gut barrier health.
Mild laxative effect: At moderate doses, this can help keep things moving, which is why glycerol and some polyols are used in gentle laxatives and in foods labeled as “high-fiber”.
So, while “too much” of any polyol can upset the stomach, their controlled use in food and medicine offers real benefits: better teeth, steadier blood sugar, and a kinder gut environment (for many, not all). They’re not miracle workers, but they’re far from “bad” alcohols.
The Grand Toast: Not All Booze Is Bad
All alcohols share that -OH swagger — hydrogen bonding for gooey viscosities, flammability for fire tricks — but chain length and OH count flip the script. Short monos? Zippy and risky. Polys? Sticky sweethearts in candy and creams. Methanol kills quietly, ethanol buzzes then bruises, glycerol nurtures. Next time you swab with isopropanol or sweeten tea with glycerol, tip your hat to the family. Some are more equal — pick wisely!
The Whiskey Maker’s Secret: Chemistry in the Still
A whiskey distiller, hands deep in grains, yeast, and oak, is in fact a practical chemist in disguise. They must know the spectrum of alcohols not just to avoid the monsters like methanol, but to craft magic, not just spirit.
Methanol: The lurking ghost — it distills early, at a lower boiling point than ethanol. A skilled distiller carefully discards the first “foreshots,” rich in methanol and other sharper volatiles, so the heart cut stays clean and safe.
Ethanol: The star — but even ethanol has phases. Too much fermentation, too fast, and off-flavors arise. Too much heat in the still, or poor control, and the alcohol burns harsh, not smooth.
Higher alcohols (fusel oils): These are the longer-chain alcohols (like propanol, butanol) that form in fermentation. In tiny amounts, they add complexity and depth; in excess, they deliver a headache and a bitter edge. The distiller rides this fine line, separating the “hearts” from the “tails” and managing aging to tame them softly.
Polyols and glycerol: These are the unsung heroes of mouthfeel. Glycerol, produced in small amounts by yeast, adds a silky, slightly sweet body to the spirit. It’s the “bubble gum” or “round” character in good whiskey, a hint that the fermentation and distillation were nurturing, not brutal.
So when the distiller cuts the run, ages the spirit, and finally bottles it, they’re not just following a recipe — they’re balancing the entire family of alcohols: the deadly, the fun, the oily, and the sweet. The deeper they understand those physical and chemical differences, the closer they get to turning grains and water into liquid poetry, not just poison or punch. Because in the world of alcohols, as in Orwell’s world, all are born equal, but only the wise can tell which ones are truly more equal than others.
Final Sip: Knowledge, Not Just Alcohol, Is Where the Power Lies
So now you know: the world of alcohols is far bigger than your local bar. Some are fierce and forbidden, some are fun and fleeting, and others are sweet, useful, and even kind. But here’s the real magic — seeing them for what they are. You’re not just drinking or using them; you’re choosing, judging, and understanding.
That’s the true armor: awareness.
A wise old chemist might say:
“To know the molecule is to master the moment. Use it gently, use it wisely, and remember — the best spirit flows not from the bottle, but from the heart.”
Now go forth: sip responsibly, clean gently, sweeten thoughtfully, and live like someone who knows that all alcohols may be born equal… but only the clever ones stay standing.
Contact
Questions or thoughts? Reach out anytime.
info@quantumsatis.site
Phone: TBD
© 2026. All rights reserved.
