The Art of Knowing When to Stop: A Guide to Fractionation
A funny excursion into fascinating problem of separation. Unveiling the elegant solution (pun intended)
CRAFTSCIENCE
7/7/202510 min read
"The beginning of wisdom is to call things by their proper names." — Chinese Proverb
My father could never leave a party at the right time. He'd arrive exactly when invited—7:00 PM meant 7:00 PM, not 7:03 — but departures were a different story entirely. We'd say our goodbyes at 9:30, and then he'd remember one more story about his time in the Navy, or he'd decide the host needed advice about their carburetor, and by 11:00 PM we'd still be standing in the doorway while my mother made the face that meant we'd be discussing this in the car.
The problem wasn't that Dad didn't know when to leave. It's that he couldn't identify the moment. The precise instant when "pleasant evening" tips into "overstaying welcome." That golden window between "they'll miss us when we're gone" and "dear God, are they ever leaving?"
Distillers face the same problem, except instead of party etiquette, they're dealing with poisonous methanol and the laws of thermodynamics.
The Deceptively Simple Problem
The whole point of distillation is separation. You heat up your fermented wash — basically beer without hops — and different liquids evaporate at different temperatures:
Methanol (the stuff that blinds you): boils at 65°C.
Ethanol (the stuff you want): boils at 78°C.
Water (the stuff that's everywhere): boils at 100°C.
So the plan seems obvious: heat the mixture to 65°C, catch the methanol, discard it. Heat to 78°C, catch the ethanol, keep it. Easy, right?


Except chemistry, like my father, doesn't respect boundaries.
The Azeotropy Problem: Or, When Molecules Become Codependent
Here's where we encounter azeotropy, which sounds like a minor European principality but is actually chemistry's way of saying "these molecules refuse to separate because they have abandonment issues."
An azeotrope is a mixture of liquids that boils as if it were a single substance. Ethanol and water are particularly codependent. When you mix them, they form hydrogen bonds — think of it as molecular hand-holding — and they refuse to let go.
The specific problem:
When you get to about 95.6% ethanol and 4.4% water, they reach what chemists call an azeotropic point. At this exact ratio, they both boil together at 78.2°C. You can't separate them further through simple distillation. They've become the couple who finishes each other's sentences and refers to themselves as "we."
"Are we going to the store?"
"We don't like asparagus."
"We boil at exactly 78.2°C."
How boiling temperature changes with alcohol percentage:
This is where it gets weird. If you track the boiling point as you change the alcohol-to-water ratio, it doesn't follow a nice straight line. It's a curve that looks like a depressed smile:
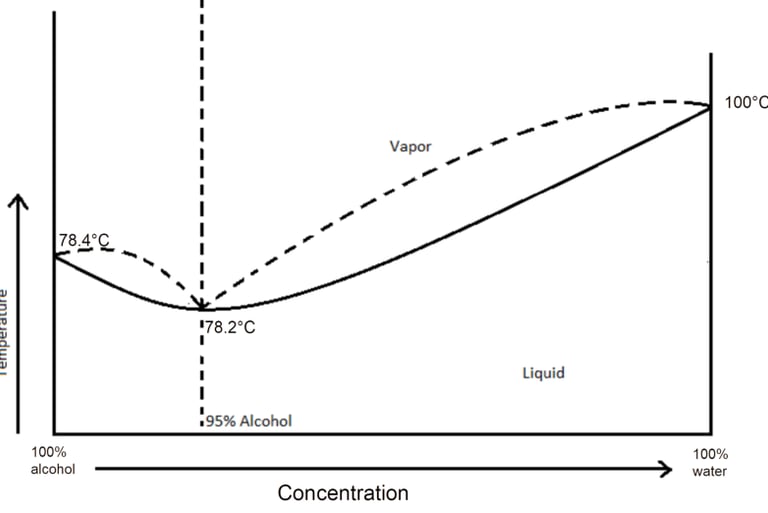
The boiling temperature decreases as you add ethanol, reaches a minimum at the azeotrope, and then... stops cooperating. You can't get to pure ethanol through heat alone. The molecules have decided this is as far as they're willing to go without couples therapy.
My aunt and uncle had a similar relationship. They argued about everything but refused to divorce because "the paperwork seemed complicated." They're still together, still arguing, still inseparable in the most dysfunctional way possible.
This is why distillers gave up trying to get pure ethanol and settled for "close enough" — which in distilling means 95% ethanol, and in life means "I'll text you when I get home" at 2AM when you both know you're not texting anyone.
Pot Stills: The Art of Manual Intervention
Pot stills are the traditional way to make whisky, rum, brandy, and anything requiring actual flavor. They're also where the art of "knowing when to stop" becomes crucial because unlike my father at parties, you can't just keep going until someone physically pushes you out the door.
Distillers make what are called cuts — decision points where they switch collection vessels to separate Heads, Hearts, and Tails. Making these cuts requires four types of information, arranged roughly in order of reliability:
A. Alcohol Strength (ABV) — The Primary Signal
The alcohol percentage in your distillate drops continuously during a run, like your enthusiasm for family reunions as you get older.
Typical ranges (these vary, but generally):
Heads: 75–85% ABV (too strong, chemically aggressive, smells like regret).
Hearts: 60–75% ABV (the good stuff, the reason you're here).
Tails: below 60% ABV (weak, oily, tastes like disappointment).
Large professional distilleries set exact thresholds: "Divert to feints tank when ABV drops below 62%." This is written in the Standard Operating Procedure, laminated, and hung on the wall like the Ten Commandments, except it's legally enforceable and actually followed.
This gives you repeatability. The same spirit, run after run, regardless of whether Bob or Susan is manning the still. Bob might be going through a divorce and Susan might be training for a triathlon, but the whiskey tastes the same because the numbers don't care about your feelings.
I once worked in an office where we had a procedure manual for making coffee. It specified exactly how many scoops per pot (2 per cup), what temperature water (90-96°C), and how long to let it brew (4 minutes). Nobody followed it. The coffee was terrible and different every day, and we all pretended this was normal.


B. Temperature — The Supporting Actor
The temperature at the still head (the top of the pot) rises during the run as heavier compounds start evaporating. Professionals use this for trend detection, not decision-making.
Temperature tells you something is changing, not what is changing or when to act.
This is important: professionals never cut by temperature alone. That's what beginners do, following online tutorials that say "cut to hearts at 78°C!" as if chemistry were that simple.
Temperature is the friend who says, "Yeah, things seem different lately" but can't tell you whether your spouse is planning a surprise party or a divorce. Useful context, but not actionable intelligence.
C. Sensory Evaluation — Still Essential Despite Living in the Future
Even with fancy instruments and digital readouts, trained distillers still:
Smell the distillate every few minutes.
Taste diluted samples.
Look for oiliness or clouding.
Generally behave like wine snobs, except they're right.
Common sensory cues:
Heads smell like:
Sharp solvents.
Green apple (but in a threatening way).
Nail polish remover.
That glue we weren't supposed to sniff in elementary school.
Hearts smell like:
Clean grain or fruit.
Sweet, pleasant alcohol.
What you hoped whiskey would smell like.
Success.
Tails smell like:
Wet cardboard left in a basement.
Bitter vegetables.
Oil.
The inside of a gym bag that hasn't been washed in a year.
Tails also have an oily mouthfeel, like someone added a tiny bit of salad dressing to your drink.
Sensory evaluation catches things instruments miss. Sometimes you get an ester spike — a sudden burst of fruity compounds that smells like artificial banana or pineapple. The ABV meter doesn't care. The temperature gauge doesn't notice. But your nose knows immediately that something weird just happened.
I have a friend who can detect a gas leak from three rooms away. She also claims to smell when people are lying, which I don't believe, but I've never actually tested her on it because what if she's right?
D. Time & Volume — For Consistency
Many distilleries define cuts based on:
Volume collected.
Elapsed time.
Historical run profiles.
A typical SOP might read:
"Heads = first 2.5% of charge volume.
Hearts until output falls below 62% ABV.
Remaining volume collected as feints".
This ensures the same spirit profile every single run, because paying customers expect their whiskey to taste like whiskey, not like a chemistry experiment gone wrong.


It's the difference between my grandmother's recipes ("add flour until it feels right") and professional bakeries ("exactly 450g flour, measured"). My grandmother's cookies varied wildly. Sometimes they were perfect, sometimes they were hockey pucks. The bakery's cookies are always the same, which is less magical but more profitable.
The Speed Problem: Or, Why Patience Actually Matters
There's a Turkish proverb that says, "Only shaitan (the devil) runs fast. Good things go slowly." It turns out this applies to distillation in ways that took me entirely too long to comprehend.
At Quantum Satis, they've discovered something that should probably be obvious but apparently isn't: the speed of heating matters as much as what temperature you reach. Not just for yield or ABV or any of the numbers we've been obsessing over — but for flavor itself.
Heat too fast and too hard, and you get what chemists call "poor separation." Multiple compounds with different boiling points evaporate simultaneously, all mixed together like a bad smoothie. Volatile esters, alcohols, and whatever else was waiting its turn all rush out at once.
It's exactly like shouting "FIRE!" in the middle of a crowded party. Everyone panics. Everyone runs for the exits at the same time. You can't tell who was leaving anyway and who just got swept up in the stampede. The careful distinction between "people who were ready to go" and "people who would have stayed another hour" gets completely lost in the chaos.
Heat slowly and gently, though, and something more controlled happens. Azeotropy develops gradually instead of all at once. The most volatile substances drift out first, then progressively less volatile compounds follow in orderly succession.
It's like if a house in the next block caught on fire during your party. People notice. They get curious. A few guests wander outside to see what's happening. Then a few more. Eventually everyone's gone, but they left one at a time, giving you the chance to say proper goodbyes, to notice who left when, to separate the "oh-I-should-probably-go" people from the "well-I-guess-the-party's-over" people.
My mother understood this instinctively when cooking. She'd make soup on the lowest possible heat setting for hours, claiming you couldn't rush flavor. My father would try to "help" by turning up the burner, thinking hotter meant faster meant better. The soup always tasted different when he "helped." Not inedible, just... hurried. Like it hadn't had time to think about what it wanted to be.
But here's the other advantage of slow heating: it gives you more time to make precise cuts.
When everything's happening fast, you're making split-second decisions. "Is this still Hearts? Or has it tipped into Tails? Quick, decide NOW!" The pressure to choose correctly while compounds are pouring out at high speed is like trying to pick the perfect wedding gift while someone holds a stopwatch and yells out how much money you're spending per second.
When you heat slowly, you have grace time. The transitions from Heads to Hearts, Hearts to Tails — they happen gradually enough that you can smell the change developing, taste the shift in character, watch the ABV creep downward instead of plummeting.
You can make the cut at the right moment instead of the panicked moment.
This is apparently what makes Quantum Satis different: they're willing to take twice as long to distill a batch if it means getting it exactly right. Which seems unremarkable until you remember that time is money, and every hour the still is running costs fuel and labor and opportunity.
Most distilleries optimize for speed. Get the batch done faster, start the next one sooner, maximize throughput. Which makes perfect business sense right up until your whiskey tastes like it was made by someone checking their watch.
It's the difference between my grandmother's hand-knitted sweaters and the ones from department stores. Both are technically sweaters. Both keep you warm. But you can tell which one was made by someone who had nowhere else to be and nothing more important to do than get every stitch exactly right.
I have one of her sweaters in my closet. It's forty years old and still perfect. The store-bought one I got last Christmas is already pilling at the elbows.
Speed has its costs. They just don't show up in the quarterly earnings report.
The Philosophy of Knowing When to Stop
The whole science of fractionation boils down to a single skill: knowing when to stop collecting one thing and start collecting another.
Stop too early: you waste good hearts by throwing them in with heads.
Stop too late: you contaminate hearts with tails.
Stop at exactly the right moment: you get consistent, high-quality spirit.
This is harder than it sounds because the transitions aren't sharp. There's no clear line where Heads become Hearts, or Hearts become Tails. It's gradual, like how your childhood imperceptibly becomes your teenage years, which blur into adulthood, and suddenly you're 40 and wondering where the time went.
The best distillers develop an intuition for these transitions. They smell the change before the instruments register it. They taste the shift in character. They've run the still so many times that they know what 63% ABV looks, smells, and tastes like without checking the hydrometer.
It's like how my mother could tell when bread was done baking without a timer — she just knew. The color, the smell, the sound it made when she tapped the crust. She couldn't explain it, and neither can master distillers, which is why training new operators takes years instead of weeks.
In Conclusion: The Azeotropic Relationship
We started with azeotropy — the annoying fact that ethanol and water refuse to fully separate because they've bonded at the molecular level.
But maybe that's not a problem. Maybe that's just reality.
Pure ethanol would be 100% efficient but also 100% terrible to drink — it would dehydrate your mouth instantly and taste like nothing except burning. The small amount of water that refuses to leave actually makes the spirit drinkable, pleasant, worth producing.
The art of distillation isn't about achieving impossible purity. It's about knowing what's good enough, when to stop, and which impurities to keep because they make the final product better.
My father never learned to leave parties at the right time. He died at 82, and his funeral was packed with people who told stories about the night he stayed until 2 AM helping them fix their water heater, or the time he missed the last train home because he was teaching someone's kid to play chess.
Maybe he was right. Maybe there's no perfect moment to leave. Maybe you just keep going until the chemistry changes, until the conversation shifts, until someone finally says "it's getting late" and you realize they're talking to you.
Or maybe I'm overthinking this, and it's just whiskey.
But whiskey is made by people who know exactly when to stop.
Contact
Questions or thoughts? Reach out anytime.
info@quantumsatis.site
Phone: TBD
© 2026. All rights reserved.
